2020-06-23
The oral microbiome is the second-largest and most diverse microbiota after the gut, harbouring over 700 species of bacteria. In The Microbiome Snapshot, we frequently cover studies of the gut microbiome since it is the largest and most diverse microbiota of the human body. However, the oral microbiome is an exceptionally complex habitat of bacteria and is the initiation point of digestion, making it crucial for maintaining oral and systematic health. [1]
The gut microbiome and oral microbiome are very different. Microbes in the mouth colonize the hard surfaces of the teeth and soft tissues of the oral mucosa. The early days of microbiome research typically used conventional culture-dependent techniques, however, the oral cavity could not be cultured. It wasn’t until next-generation sequencing, advances in bioinformatics, and effective oral microbiome collection methods emerged that the complexities of the oral microbiota could be studied.[1]
The salivary microbiome has its own characteristic microorganisms that interact with other microbiomes in the human body, especially the intestinal tract. [2]
“There is an excellent genera diversity in the human salivary microbiome; therefore, it is essential to understand the role of these known and unknown genera in the oral cavity and how they interact with the microbiome of other systems in the human body.” – Al-Zyoud et al (2020). [2]
With easy, high quality, collection devices, such as OMNIgene·ORAL, collecting salivary microbial samples for high-throughput sequencing is now possible.
Case study: Smoking tobacco and oral microbiome
A recent study from Jordan by Al-Zyoud et al (2020) called, “Salivary microbiome and cigarette smoking: A first of its kind investigation in Jordan,” was recently published regarding the effects of smoking on the oral microbiome.[2]
The oral microbiota is essential in inducing a dynamic equilibrium with the immune-inflammatory response of the human host.
“The oral cavity is one of the entries of the respiratory tract, and the main entry point for several microorganisms, primary airborne pathogens, and those transferred through saliva.” – Al-Zyoud et al (2020).
Tobacco and the salivary microbiota
Tobacco smoking generates carcinogens that contain distinct nitrosamines and free radicals capable of inhabiting antioxidant enzymes.
“An inhabited antioxidant enzyme makes the oral epithelial cells unprotected against the damaging effects of thiocyanate ions and hydroxyl free radicals. Thiocyanate ions and free radicals could react with DNA, adversely, therefore, open the gateway to the progression of oral cancer. By inhibiting granulocyte function, smoking impairs host defenses and affects the immune system.” – Al-Zyoud et al (2020).
Smoking contributes to the increase in the number of neutrophils in peripheral blood. These changes result in activating inflammatory cells, which leads to the release of free radicals, influencing DNA damage.[2]
Al-Zyoud et al (2020), collected saliva samples using OMNIgene·ORAL (OM-501) from 100 subjects to learn more about the effects of tobacco on the salivary oral microbiome. Fifty-one subjects were non-smokers and 49 were smokers.
[Note: the OMNIgene·ORAL OM-501 only collects microbial DNA from saliva. It is also possible to collect microbial DNA and RNA from saliva using the OMNIgene·ORAL OM-505. Click here for more details.]
They ran the saliva samples on high throughput 16S rRNA sequencing and were successful in generating high-quality sequencing data that enabled the analysis and identification of the significant compositional difference between smokers and non-smokers.[2]
Salivary oral microbiota of smokers versus non-smokers
Based on their results, Al-Zyoud et al (2020), found quite a difference within the oral microbiota between the smokers versus non-smokers. Figure 4 and 5 from their paper below, shows the taxonomic differences between smokers and non-smokers.
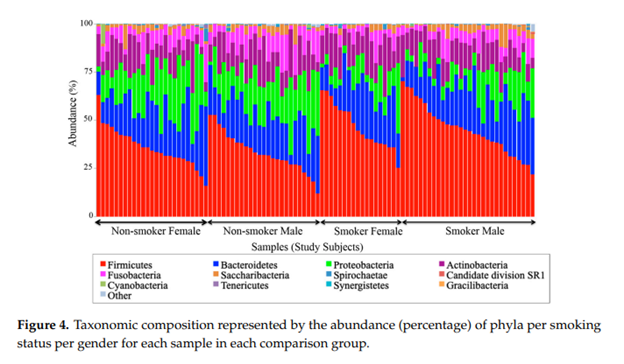
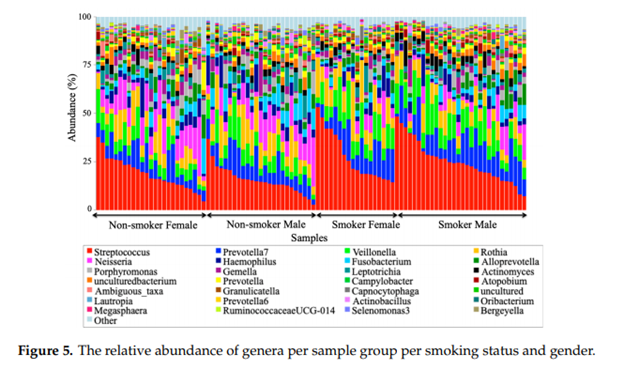
The study showed that:
- Smoking had affected the Firmicutes, Proteobacteria, and Fusobacteria.
- Firmicutes were statistically elevated in smokers at the expense of Proteobacteria and Fusobacteria in non-smokers.
- According to the authors, this implies that smoking has a critical impact on the homeostasis of the human salivary microbiome. The biological meaning of these findings was not evident until they performed the analysis at the genera level (seen in figure 5 above).
- Smokers had a reduced level of Neisseria which might be related to the effect of oxygen deprivation in the oral cavity caused by smoking.
- Smokers also had increased levels of Streptococcus and Veillonella which may explain their success in tolerating the lack of oxygen in the smoking microenvironment. [2]
“We can suggest that there is a microbial signature at the genera level that can be used to classify smokers and non-smokers [based on] the salivary abundance of the 15 genera including, but not limited to, Streptococcus, Prevotella, and, Veillonella, which are all more abundant in smokers relative to non-smokers, and Neisseria, which is more abundant in non-smokers relative to smokers.” – Al-Zyoud (2020).
With the combination of a collection method providing a high quality sample, accurate sequencing and advanced bioinformatics, Al-Zyoud et al (2020), were able to prove the impact that smoking has on the core salivary microbiome, causing a significant shift in the salivary microbiome in smokers.
If you are interested to learn more about collecting salivary microbiome, you can email us at info@dnagenotek.com or click the button below to request free trial kits for your study. We also offer microbiome lab services through our sister company Diversigen.
Related blog
Innovation to facilitate oral microbiome studies
References:
[1] Priya Nimish Deo and Revati Deshmukh. Oral microbiome: unveiling the fundamentals. J Oral Maxillofac Pathol. 23(1):122-128 (2019).
[2] Al-Zyoud et al. Salivary Microbiome and Cigarette Smoking: A First of Its Kind Investigation in Jordan. Int. J. Environ. Res. Public Health.17:256 (2020).


